 
Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. 
Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. This is, after all, how we produce TV signals. 
It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. It is named after Niels Bohr, due to its role in the Bohr model of an atom. h E ( 1 n l o w 2 1 n h i g h 2) 13.6 eV. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. 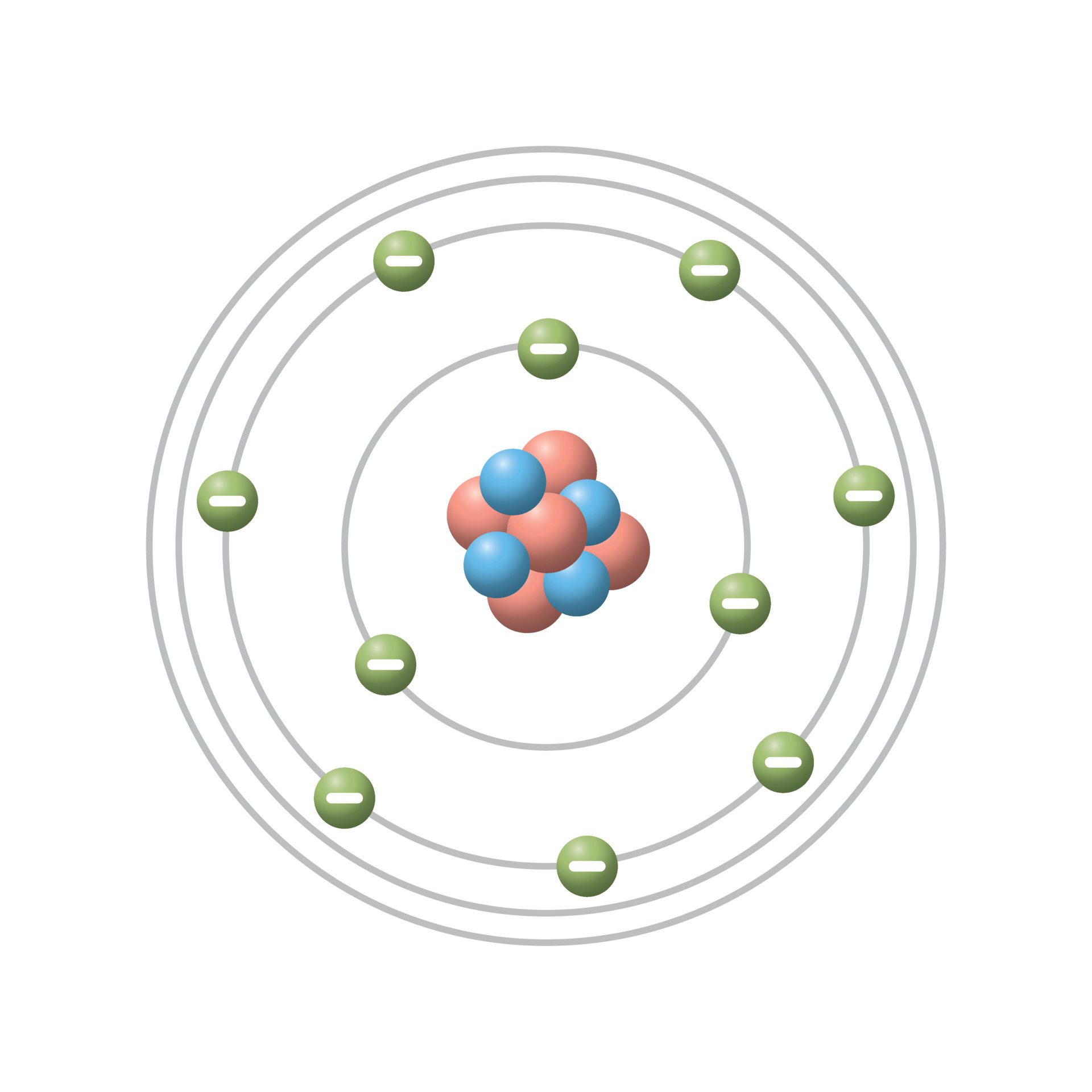
Bromine is neutral and its atomic number is 35, hence, the number of protons and electrons available for its Bohr diagram. The Bohr radius ( ) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Bohrs model calculated the following energies for an electron in the shell, n. Unfortunately, there was a serious flaw in the planetary model. Quantum mechanics emerged in the mid-1920s. The Bohr model of Bromine (Br) is drawn with four electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons, the third shell contains 18 electrons and the fourth shell contains 7 electrons. Bohrs model suggests that each atom has a set of unchangeable energy levels, and electrons in the electron cloud of that atom must be in one of those energy levels. For a more in-depth discussion of the history of atomic models, see atom: development of atomic theory. 2.12: The Bohr Model - Atoms with Orbits is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew. Atomic models have gone through many changes over time, evolving as necessary to fit experimental data. \) for a summary).\): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). Learn about the Bohr model of the atom and how it explains emission and absorption spectra of elements. Atomic model, in physics, a model used to describe the structure and makeup of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
